Adverse reactions reported on corona vaccines
Updated 7 March 2024
| Type of vaccine (product name) | Total number of reports** | Non-serious** | Serious*,** |
|---|---|---|---|
| mRNA vaccine Comirnaty (including variant vaccines) | 19809 | 14825 | 4984 |
| mRNA vaccine Spikevax (Moderna) | 5675 | 4571 | 1104 |
| Adenovirus vector vaccine Vaxzevria (AstraZeneca) | 2973 | 2060 | 913 |
| Adenovirus vector vaccine COVID-19 Vaccine Janssen | 14 | 9 | 5 |
| Protein-based vaccine Nuvaxovid (Novavax) | 27 | 18 | 9 |
| Total | 28498 | 21483 | 7015 |
* Seriousness assessment is based on reporter’s assessment, or if not available, on Fimea’s assessment. An adverse reaction is considered serious if it is fatal or life-threatening, requires hospitalisation or prolongs it, causes persistent or significant disability or incapacity or congenital anomaly.
** The total number may be less than the sum of the different vaccines, as one report may contain more than one corona vaccine as a suspected vaccine.
Finnish Medicines Agency Fimea has a webpage that provides information on the adverse reaction reports submitted for corona vaccines. The page is updated once a month with the number of submitted reports and a more detailed report in Finnish.
How to report a suspected adverse reaction
An adverse reaction reporting system is a pharmacovigilance tool. The main objective of the system is to detect possible new adverse reactions. Healthcare professionals and consumers are asked to report adverse reactions that are:
- unexpected – i.e. they are not listed in the package leaflet of the vaccine
- serious – i.e. they are fatal or life-threatening, require hospitalisation or prolong it, cause persistent or significant disability or incapacity (e.g. long-term incapacity for work) or congenital malformation.
It is not necessary to report adverse reactions that are identical with those described in the package leaflet (e.g. transient fever, muscle pain, joint pain or injection site pain).
Adverse reaction reports can be submitted by healthcare professionals and consumers. The report should include the product name of the vaccine, and if possible, the batch number that can be found in My Kanta online service.
Further information: Submitting a report on an adverse reaction (Fimea)
What happens to adverse reaction reports
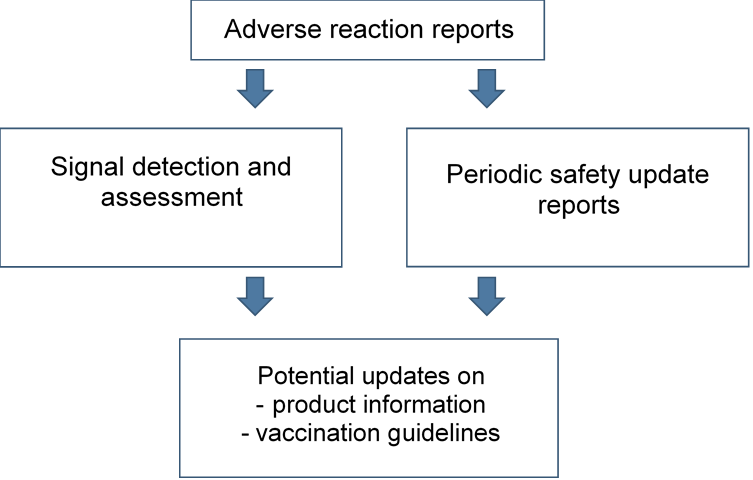
All adverse reaction reports on corona vaccines are stored in Fimea’s adverse reaction register and sent regularly to Finnish Institute for health and welfare. In addition, all reports are sent to EudraVigilance database, which is maintained by the European Medicines Agency (EMA). The vaccine marketing authorisation holders and World Health Organisation (WHO) have access to adverse reaction reports via EudraVigilance. Also consumers can view the reports in the database.
The electronic databases are searched for signals, i.e. signs of potential new adverse reactions. Conclusions on the benefits and risks of vaccines cannot be made until all available information has been scientifically evaluated. The adverse reaction reports are also assessed in Periodic Safety Update Reports (PSURs) that review all information received about vaccines and the benefit-risk balance.
Further information: Adverse reactions (Fimea)