National clinical trial applications
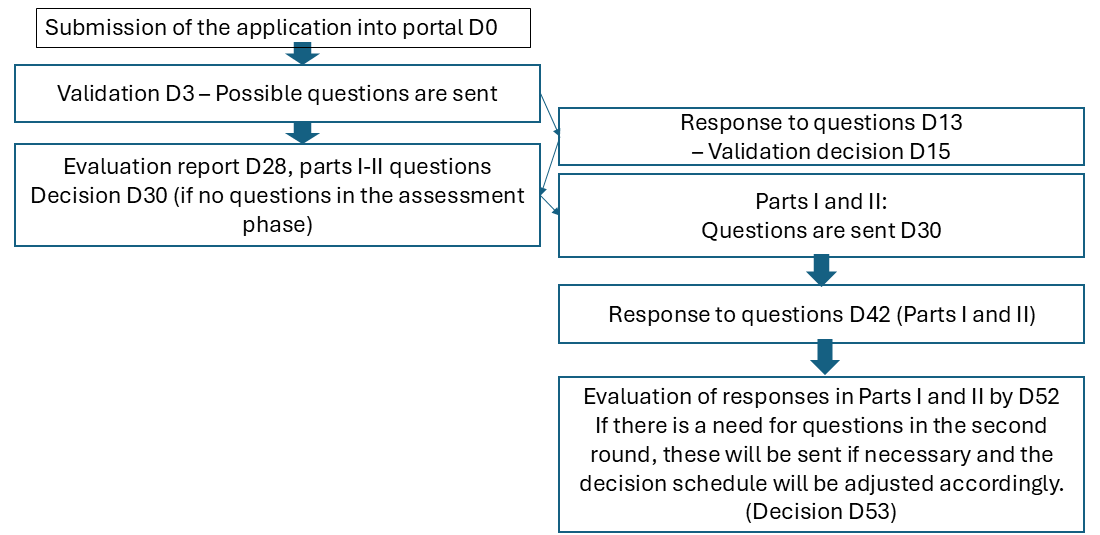
For national clinical trial applications sent only to Finland, an accelerated evaluation schedule is followed, with a fixed schedule for different phases. The progression of the schedule starts from the arrival of the application in the CTIS portal, and progression is not affected by issues such as questions in the validation phase.
Friday is not recommended as the date of submission of the application. If the application is sent to the portal on a Friday, during a weekend or on a midweek holiday, the delivery date is calculated as the next working day (D0). Application handling fees, obligations and other activities remain the same compared to multinational studies. See "Trial application and modifications".
Below is a table describing the processing phases for the application sent to the portal, according to what day of the week is in question. Week 1 is the week when the application is submitted. If the sponsor’s response arrives on a midweek holiday, the portal will not delay this. This should be taken into account in the trial application submission schedule.
| Phase | Monday | Tuesday | Wednesday | Thursday | Friday |
|---|---|---|---|---|---|
| Validation questions will follow | Week 1, Thursday | Week 1, Friday | Week 2, Monday | Week 2, Monday | Week 2, Thursday |
| Answering the validation questions | Week 3, Monday | Week 3, Monday | Week 3, Thursday | Week 3, Thursday | Week 4, Monday |
| Answering the validation questions | Week 5, Wednesday | Week 5, Thursday | Week 5, Friday | Week 6, Monday | Week 6, Wednesday |
| Evaluation questions arrive/ decision if no questions | Week 7, Monday | Week 7, Tuesday | Week 7, Wednesday | Week 8, Monday | Week 8, Monday |
| Evaluation questions processed | Week 8, Thursday | Week 8, Friday | Week 9, Monday | Week 9, Thursday | Week 9, Thursday |
| Decision if evaluation questions have been sent | Week 8, Friday | Week 9, Monday | Week 9, Tuesday | Week 9, Friday | Week 9, Friday |
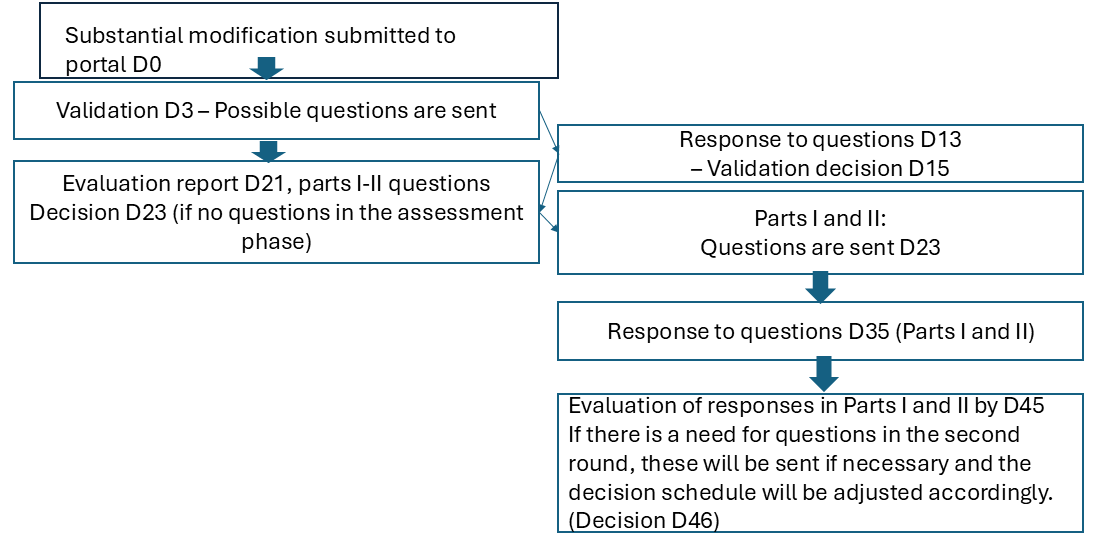
Substantial modifications in national application processes
In accordance with the accelerated schedule, the clinical trial applications whose initial assessment has also been accelerated will be processed accordingly--in other words, this accelerated process will apply to applications initiated on 11 February 2026. Other applications will be processed according to the normal CTIS portal schedule. In case of substantial modifications, the progression of the schedules works in the same way with respect to the submission of the substantial modifications application to the portal. The table for initial applications is otherwise valid, but the evaluation questions and the subsequent process take place one week earlier.